A GAMP4 compliant software platform that automates chemotherapy and TPN production with real-time inventory and compliance management.
What We Did
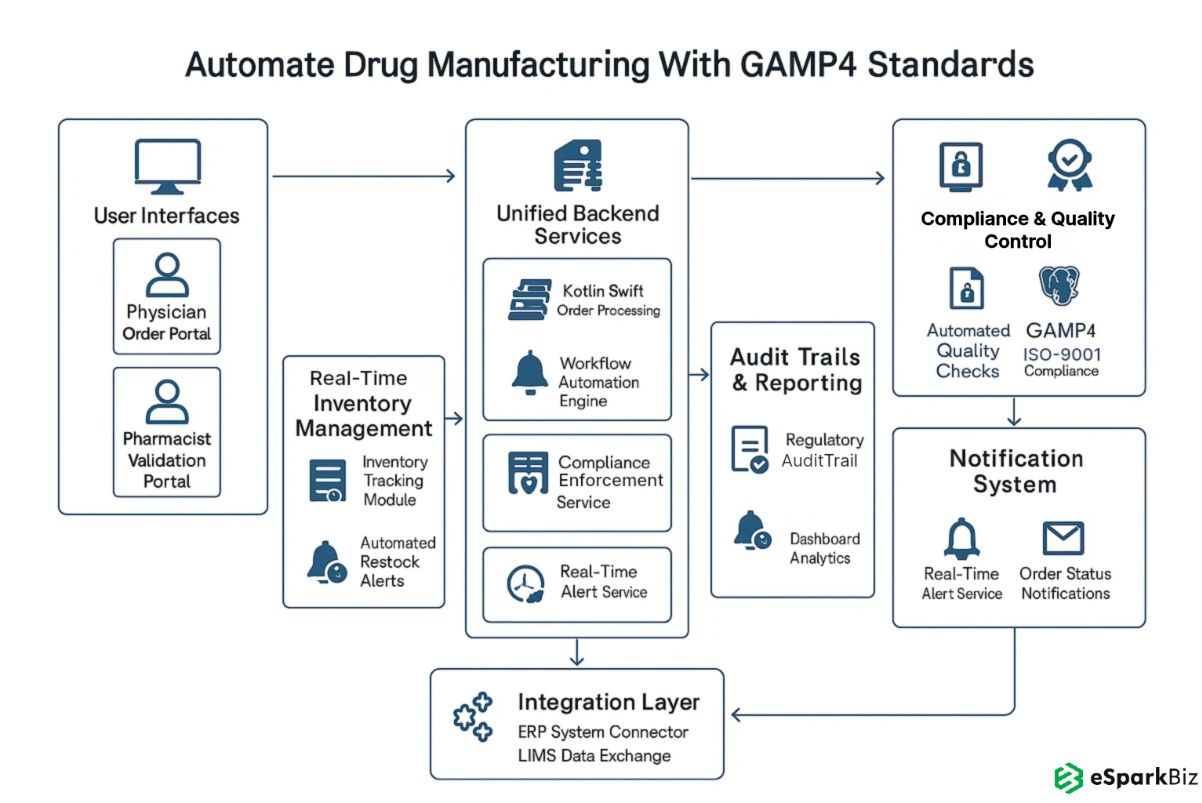
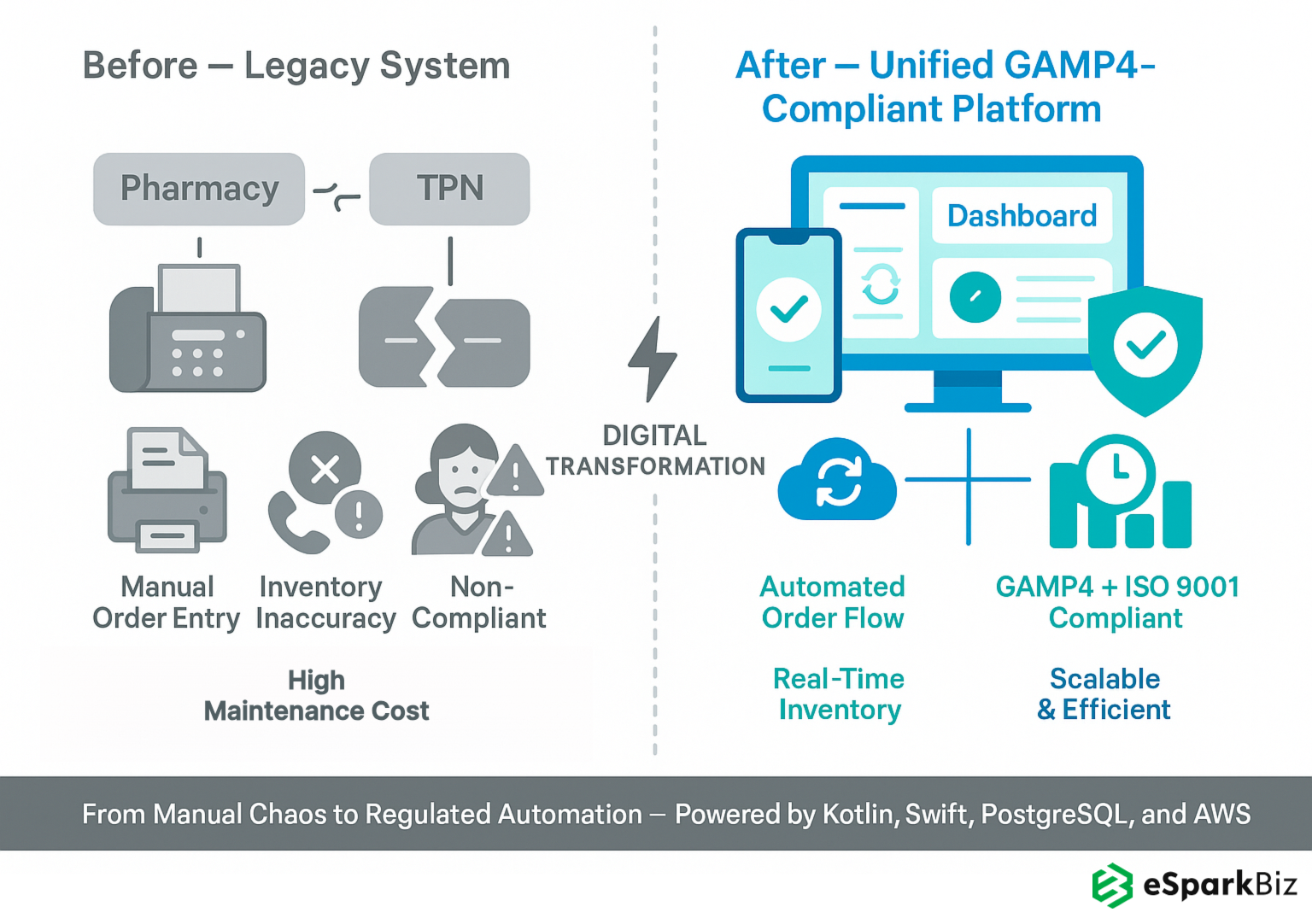
- Built a unified, GAMP4 compliant platform to automate chemotherapy and TPN production, replacing multiple legacy systems.
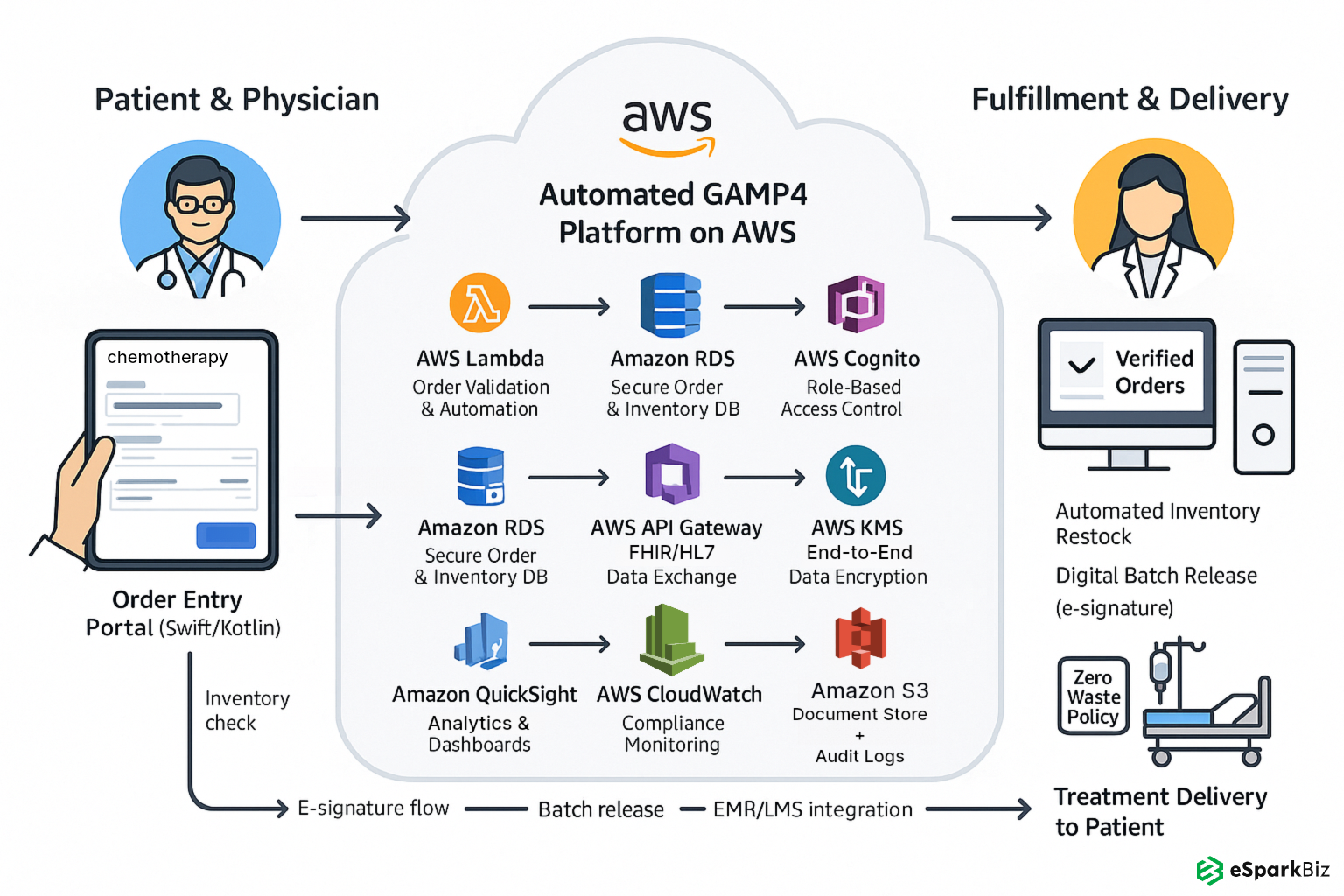
- Created mobile ready interfaces using Kotlin and Swift so pharmacy and clinical teams can access in real time.
- Implemented real time inventory tracking and auto restock, 99.9% stock accuracy and no outages.
- Digitized QA and batch release with e-signatures and audit trails, reduced approval times and regulatory compliance.
- Integrated with hospital EMRs and lab LIMS via HL7/FHIR APIs for seamless data exchange and treatment accuracy.
- Enforced role based access controls (RBAC) and end to end data encryption to protect patient data and meet HIPAA, ISO-9001 and GAMP4 standards.
GAMP4 System for a Large Pharmaceutical Company
They built a unified platform using Kotlin, Swift and PostgreSQL to automate chemotherapy and TPN production, replacing a siloed system and meeting strict regulatory requirements. It ensured data integrity across the board.
The diagram illustrates a GAMP4-compliant system streamlining drug manufacturing, enhancing compliance, automation, and real-time order processing:
Simplifying the Manufacturing Process
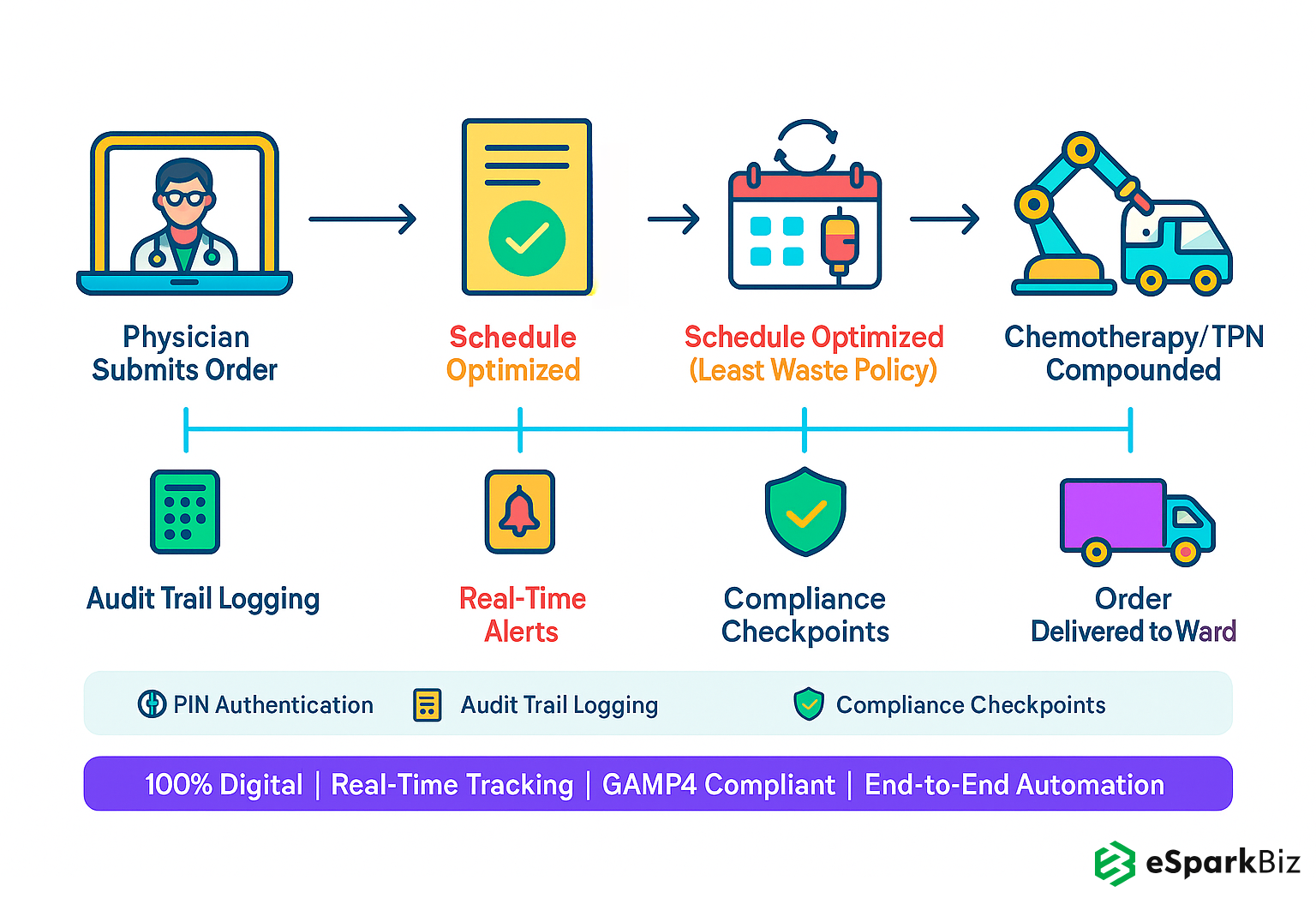
They automated order processing from physician submission to delivery, validated each order automatically and optimized multi-dose schedules, reduced order-to-delivery time by 40% and waste with a least-waste policy. This saved cost big time.
Real Time Inventory and Automated Restocking
They implemented real time inventory monitoring and automated restock alerts, maintained 99.9% stock accuracy and no stockouts, kept production running smoothly and eliminated stockouts. This improved resource utilization.
Invoice Management
They built quick-create, search and filter for invoices, reduced billing time by 60% and administrative work. Teams processed invoices faster every day, simplified finance process and overall productivity.
In-Depth Analytics for Decision Making
They added KPI dashboards and reports to track production, waste and quality metrics, enabled managers to make data driven decisions and improve operational efficiency across all departments. This brought proactive management.
Data Integrity and Regulatory Compliance
They implemented PIN authentication and automated quality checks to ensure 100% GAMP4 compliance, patient data and audit trails, product quality and regulatory standing. It minimized compliance risk.
💡 Did you know?
The global pharmaceutical manufacturing software market is expected to grow from $3.3 billion in 2024 to $3.82 billion in 2025, a 16% increase driven by increasing adoption of automation and compliance solutions.